Introduction
In many laboratories, PCR plates are treated as standardized consumables. As long as the format matches the instrument—96-well, 384-well, standard footprint—they are usually assumed to be interchangeable without much further evaluation.
In practice, that assumption is not always reliable. A plate can fit into the thermal cycler, allow the lid to close normally, and still behave differently once the workflow begins. The reactions may amplify without obvious failure, but the agreement between wells can start to weaken. This is often where compatibility becomes difficult to recognize: the issue does not look like a broken process, but like a gradual loss of consistency.
When this happens, familiar explanations are usually checked first. Pipetting technique, reagent quality, cycling conditions, and sealing method are all reasonable places to start. But if the same type of variation remains after these factors have been controlled, it becomes worth asking whether the plate is interacting with the thermal cycler in a fully consistent way.
In plate-based PCR and qPCR workflows, compatibility is not only about whether a plate physically fits the instrument. It also depends on how uniformly the plate maintains contact, alignment, and pressure across all wells during real use. This becomes especially important in low-volume reactions and qPCR applications, where small physical differences are more likely to appear in the data.
Understanding PCR plate compatibility in this way gives laboratories a more practical route for troubleshooting. Instead of treating the plate as a neutral container, it helps to evaluate whether the plate, instrument, and sealing system are working together as a stable physical system.
Compatibility Is Not Just About Footprint
In most workflows, compatibility is often judged by whether a PCR plate matches the standard footprint of the instrument. If the plate fits into the thermal cycler and the lid closes without resistance, it is usually considered suitable for use. From a practical standpoint, this is reasonable. Dimensional compatibility is the first requirement, and in many cases, it works well enough.
However, this is also where a common assumption begins to form—that plates with the same format can be used interchangeably without affecting performance. In practice, plates that share the same external dimensions do not always behave the same way once they are used in the same instrument.
These differences are rarely obvious during setup. The plate sits in place, the run starts normally, and there are no immediate signs of incompatibility. The distinction tends to appear only when results are compared more closely. Replicate wells may show slightly wider dispersion than expected, or results may vary subtly from run to run even when the protocol has not changed.
What makes this difficult to recognize is that there is no clear point of failure. The plate fits, the instrument runs, and amplification proceeds as expected. Because of this, compatibility is often reduced to a question of dimensions alone, while other factors—such as how consistently the plate maintains contact with the thermal block or how it responds under lid pressure—are not examined directly.
In practice, this means that two plates with similar specifications may perform differently under the same conditions. The difference is not in whether they can be used, but in how consistently they support uniform reaction conditions across all wells. This becomes more noticeable in workflows where small variations matter, such as low-volume reactions or qPCR experiments.
For laboratories evaluating different formats, choosing the right PCR plate for specific applications often provides a more reliable basis for comparison than relying on nominal compatibility alone.
Thermal Block Contact Defines Reaction Consistency
Once a plate is placed into a thermal cycler, it is easy to assume that all wells experience the same heating conditions. The block is designed to provide uniform temperature control, and the cycling program is applied equally across the entire plate. In most cases, this assumption is sufficient and does not raise concerns.
However, uniform temperature control depends on one condition that is often taken for granted: consistent contact between the plate and the thermal block.

In practice, this contact is not always perfectly uniform. Small differences in plate flatness, rigidity, or how the plate settles into the block can lead to slight variations in heat transfer between wells. These differences are usually subtle. They do not prevent amplification and rarely trigger any warning from the instrument.
What they influence is how evenly reaction conditions are maintained across the plate. When heat transfer varies slightly between wells, reaction environments begin to diverge. This is more likely to become noticeable in low-volume reactions or qPCR workflows, where small thermal differences can affect how closely replicates align. In practice, a Ct standard deviation above approximately 0.3–0.5 between technical replicates may begin to indicate that reaction conditions are not being maintained uniformly, particularly when the variation follows a consistent spatial pattern across the plate.
One of the reasons this is difficult to identify is that temperature-related variation is often attributed to the thermal cycler itself. When inconsistencies appear, the instrument is usually the first variable to be questioned. But if the block is operating within specifications, the difference may lie in how effectively the plate is maintaining uniform contact across its surface.
In routine workflows, this is rarely evaluated directly. Plates are not typically assessed based on how evenly they sit in the block, and small differences are easy to overlook during setup. As a result, variation introduced at the level of thermal contact can persist across runs without being clearly recognized as a separate factor.
Heated Lid Pressure Interacts with Plate Structure
In most thermal cyclers, the heated lid is designed to apply consistent pressure across the entire plate. This pressure serves a practical role. It helps maintain close contact between the plate and the thermal block, while also supporting sealing by pressing the film or cap system evenly across all wells.
Under ideal conditions, this pressure is distributed uniformly. In practice, however, how that pressure is actually experienced across the plate depends not only on the instrument, but also on the plate itself.
Different plates respond to lid pressure in different ways. Variations in rigidity, thickness, or structural support influence how force is transmitted across the plate surface. A more rigid plate tends to maintain its shape under pressure, allowing contact to remain relatively even. A more flexible plate may deform slightly when the lid is engaged, causing pressure to be redistributed across different regions.
These differences are usually small and not visible during setup. The lid closes normally, and there is no indication that anything is misaligned. The effect tends to become noticeable only during thermal cycling. Wells that receive slightly less pressure may not maintain the same level of contact or sealing performance as others, while areas under more consistent pressure tend to behave more uniformly.
This is more likely to be observed in low-volume reactions or qPCR workflows, where small differences in evaporation control or contact conditions can influence how closely replicates align. In practice, even subtle variations in how the plate is sealed can contribute to measurable differences in consistency, particularly when pressure is not distributed evenly across the plate.
What makes this difficult to interpret is that the instrument itself is functioning as intended. The lid applies pressure as designed, and from an operational perspective, everything appears correct. The variation arises from how the plate structure interacts with that pressure, rather than from any failure of the system.
In practice, this interaction is rarely evaluated directly. When differences in results appear—especially those that seem related to sealing or evaporation—they are often attributed to how the seal was applied, rather than to how pressure was distributed across the plate. As a result, the role of plate structure in shaping pressure distribution is easy to overlook, even though it directly influences how uniformly conditions are maintained across wells. For a more detailed discussion of how sealing performance influences PCR and qPCR consistency, see how plate sealing affects PCR and qPCR reliability.
Plate Geometry Influences Alignment Before Cycling Begins
Before a PCR run even starts, a series of small handling steps have already taken place. Plates are positioned on the bench, filled, sealed, and often moved between workstations before being placed into the thermal cycler. These steps are routine and usually treated as neutral, with the assumption that they do not significantly influence the outcome of the experiment.
In real workflows, this assumption does not always hold. The way a plate behaves during handling can influence how consistently it is aligned and supported by the time it reaches the thermal cycler.
Plate geometry plays a role here. Differences in structural support—such as how rigidity is distributed around the edges or how evenly the base is reinforced—can affect how stable the plate remains during routine handling. Some plates maintain their shape more consistently, while others may flex slightly when lifted, sealed, or repositioned. These changes are typically small and not noticeable during normal operation.
However, even minor variations in alignment can carry forward into the thermal cycling step. If a plate does not sit completely flat when it is placed into the block, or if its position shifts slightly during handling, the initial contact conditions are already uneven before the lid is closed. Once cycling begins, these small differences interact with thermal contact and lid pressure, making their effects more apparent during the run.
This is more likely to be observed in workflows where handling is more frequent or less controlled, or in low-volume reactions where small differences are more easily reflected in the results.
What makes this stage easy to overlook is that nothing appears out of place. The plate fits into the cycler, sealing looks intact, and the workflow proceeds as expected. Because of this, variation that originates from earlier handling steps is often attributed to later stages, where it becomes more visible in the data.
In routine laboratory work, alignment is rarely evaluated explicitly. As long as the plate can be placed into the instrument without resistance, it is generally assumed to be positioned correctly. In reality, how the plate arrives in that position—after filling, sealing, and transport—can influence how uniformly conditions are maintained during the run.
Compatibility Issues Often Appear Only During Real Workflows
One of the reasons compatibility issues are often overlooked is that they rarely present themselves in a clear or immediate way. During initial use, most plates appear to work without any obvious problems. The instrument runs normally, amplification proceeds as expected, and there are no visible signs of misfit or failure.
In many cases, the first few runs do not raise concerns. Results fall within an acceptable range, and any small differences are easily attributed to routine variation. Because of this, compatibility is often assumed rather than verified, especially when the plate meets standard dimensional requirements.
The distinction tends to become noticeable only with repeated use or closer comparison. As more data accumulates, certain patterns begin to emerge. Replicates that are expected to align closely may show slightly wider dispersion, or results may drift subtly between runs without any clear change in protocol. These differences are usually small, but consistent enough to prompt further investigation.
What makes this difficult to interpret is that the system still appears to be functioning correctly. There is no clear failure point to investigate. Rather than a breakdown in performance, what appears is a gradual loss of agreement between wells or between runs. Under these conditions, troubleshooting tends to focus on variables that are easier to adjust—such as reagents, pipetting technique, or cycling parameters—while the interaction between the plate and the instrument is rarely examined early on.
Another factor is that these effects are condition-dependent. They tend to become more visible in low-volume reactions, high-throughput setups, or qPCR workflows, where small differences are more easily reflected in the data. In less sensitive conditions, the same plate may appear to perform adequately, reinforcing the assumption that compatibility is not an issue.
As a result, compatibility problems are often recognized only after multiple adjustments fail to resolve the variation. In some cases, the turning point comes when a different plate is introduced and the overall pattern changes. When that happens, it becomes easier to associate the effect with the plate itself, even though the underlying mechanism may still not be immediately clear.
Common Signs That Your PCR Plate May Not Fit Your Instrument Properly
In routine PCR and qPCR work, compatibility issues rarely present as obvious failures. Reactions still amplify, and there are no clear errors during setup or cycling. Instead, the signs tend to appear in more subtle ways, often becoming noticeable only when results are compared across wells or across runs.
One common indication is the presence of repeatable patterns that do not align with how the experiment was set up. For example, certain regions of the plate may consistently show higher dispersion than others, even when samples and reagents are prepared under the same conditions. When this type of pattern appears repeatedly, it is unlikely to be caused by random pipetting variation.
Another signal is persistent disagreement between replicates despite controlled handling. When pipetting technique has been checked and reagents are consistent, but replicate wells still show unexpected spread, it is more likely that reaction conditions are not being maintained uniformly across the plate. In this situation, the variation is less likely to be random and more likely to reflect how conditions are being maintained across the system.
Changes observed after switching plates can also be informative. If the overall pattern shifts when a different plate is used—while the protocol and instrument remain unchanged—it strongly suggests that the plate is influencing how conditions are maintained during the run. In practice, this is one of the more direct ways to identify compatibility-related effects.
In qPCR workflows, these signals are often easier to detect. They may appear as increased dispersion in Ct values, subtle differences in amplification curves, or reduced agreement between technical replicates. For example, consistent edge-to-center differences approaching or exceeding ~0.5 Ct often warrant closer examination, particularly when they persist across runs under the same conditions. These effects are usually small, but they can influence how confidently results are interpreted, especially in experiments that rely on precise comparisons. For a broader discussion of how plate design can contribute to variability, see why your PCR results vary: the overlooked impact of PCR plate design.
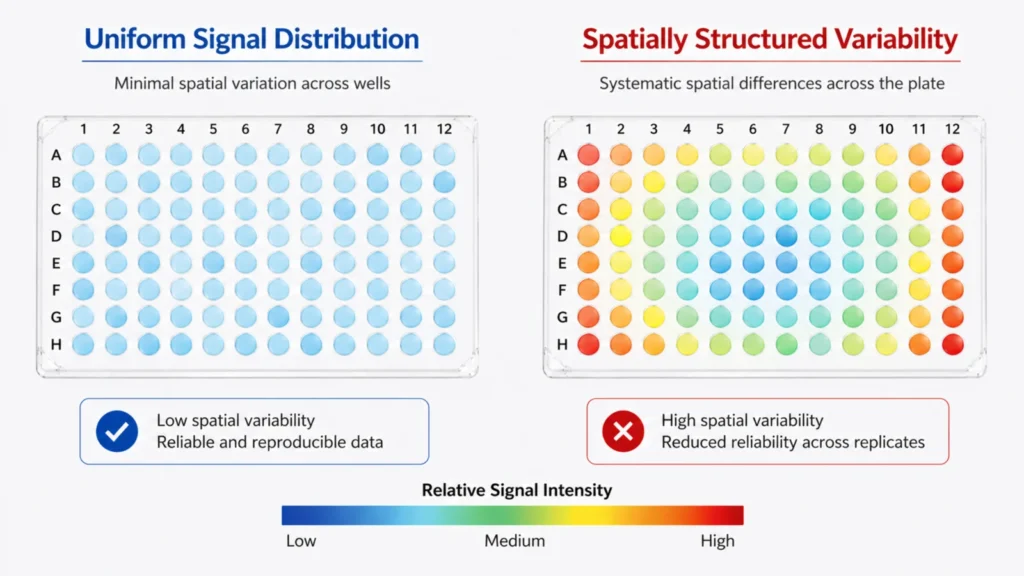
Another useful indicator is stability over time. Compatibility-related variation tends to remain consistent once it appears. Unlike random variation, which changes from run to run, these patterns are often reproducible under the same conditions. When variability is both structured and repeatable, it is more likely to reflect a system-level factor rather than an isolated setup issue.
Practical Checks Before Switching or Purchasing PCR Plates
When compatibility becomes a possible factor, the most effective approach is not to rely on specifications alone, but to observe how the plate behaves under actual workflow conditions. In many cases, simple comparisons provide clearer answers than repeatedly adjusting other variables.
A useful starting point is to evaluate how consistently the plate sits in the thermal cycler. This is not typically measured directly, but it can be inferred from result patterns. If variation follows a consistent spatial distribution across the plate, it is more likely to be related to contact or alignment than to random setup variation, even when the plate appears to fit correctly during setup.
A simple physical check can also be informative. Placing an empty plate on a flat surface and gently pressing different corners can sometimes reveal slight flexing or instability that may not be obvious during normal handling. While this does not replace in-run evaluation, it can provide a quick indication of whether the plate maintains its shape consistently.
It is also helpful to consider how results reflect sealing performance. Rather than focusing only on whether the seal is intact, it is more informative to look at whether wells behave uniformly after cycling. If certain regions consistently show different behavior, it may indicate that pressure distribution was not uniform across the plate.
Direct comparison remains one of the most practical ways to evaluate compatibility. Running the same protocol with a different plate—while keeping the instrument and setup unchanged—can quickly clarify whether the plate is influencing the outcome. If the overall pattern changes with the plate, compatibility becomes a likely contributing factor.
Observing how variation behaves over time can also provide useful insight. If the pattern remains stable across repeated runs under the same conditions, it is less likely to be random. In this case, it becomes more reasonable to consider system-level factors, including how the plate interacts with the instrument.
In qPCR workflows, generating a plate heatmap of Ct values can provide a quick visual indication of spatial patterns, helping distinguish between random variation and plate-related effects. This type of visualization is often one of the most direct ways to confirm whether observed differences follow a structured distribution.
When selecting new PCR plates, it is often worth validating performance under real operating conditions before scaling up. Reviewing different PCR plate options for specific workflows can also help ensure that plate design, rigidity, and compatibility align with the requirements of your application. Even plates that meet standard specifications may behave differently in a given workflow. A small initial evaluation can help identify potential issues early, before they become more difficult to trace.
Conclusion
PCR plate compatibility is often understood in terms of whether a plate can be used with a given instrument. In practice, however, the more relevant question is not whether a plate fits, but whether it interacts with the system in a consistent way across all wells.
As plate-based workflows become more sensitive—particularly in low-volume reactions and qPCR applications—even small differences in how physical conditions are maintained can influence how reliably results align. These effects are rarely caused by a single factor in isolation. Instead, they emerge from how the plate, thermal cycler, and sealing system function together under real operating conditions.
For this reason, evaluating compatibility is less about comparing specifications and more about observing performance in context. Plates that appear similar on paper may behave differently in practice, and these differences are often only recognized when consistency becomes a limiting factor.
Recognizing compatibility as a system-level characteristic helps shift troubleshooting away from repeated adjustments of familiar variables and toward a more complete understanding of how conditions are maintained across the plate. In many cases, this perspective provides a more direct path to improving reproducibility.
For laboratories comparing different PCR plate configurations, small differences in fit, structure, and interaction can become meaningful over repeated use. Reviewing how plates perform within specific workflows often provides a more reliable basis for selection than relying on nominal compatibility alone.
📩 For pricing information or configuration consultation, please contact us.
FAQ
Are all PCR plates compatible with any thermal cycler?
Not necessarily. Most PCR plates follow standard footprint dimensions and will physically fit into common instruments. In practice, compatibility depends on how consistently the plate maintains contact and alignment within a specific thermal cycler. Plates that fit without issue may still perform differently during cycling, particularly in terms of heat transfer and pressure distribution across wells.
Why can the same PCR plate perform differently in different instruments?
Thermal cyclers differ in how they apply lid pressure, support the plate, and maintain alignment during cycling. A plate that performs consistently in one instrument may behave differently in another if these conditions are not distributed in the same way. In most cases, the difference reflects how the plate and instrument interact as a system, rather than a problem with either one alone.
Can compatibility issues affect results even if PCR amplification appears normal?
Yes. Amplification can proceed as expected while agreement between wells begins to weaken. This is more noticeable in qPCR workflows, where small differences in reaction conditions can appear as increased Ct variation or reduced alignment between replicates.
How can I tell if variability is caused by compatibility rather than pipetting or reagents?
A practical way to distinguish this is to look for repeatable patterns. If variation appears consistently in the same regions of a plate, or persists across runs despite controlled pipetting and reagents, it is unlikely to be random. In these cases, it is more reasonable to consider whether conditions are being maintained uniformly across the plate.
What is the most practical way to evaluate PCR plate compatibility?
The most direct approach is to compare plates under the same conditions. Running the same protocol while changing only the plate allows you to observe whether the overall pattern of variation shifts. If the pattern changes with the plate, compatibility is likely to be a contributing factor.


