Introduction
In many PCR and qPCR workflows, inconsistent results are often traced back to familiar explanations—reagent quality, pipetting variation, or thermal cycler performance—the usual suspects in almost every troubleshooting discussion. These factors are undoubtedly important, but in practice, many laboratories find that even after optimizing them, variability can still persist without a clear explanation, appearing between runs, between plates, or even across wells within the same experiment.
One variable that is frequently overlooked is the PCR plate itself. Because PCR plates are treated as standardized consumables, their role is rarely questioned once a workflow is established. However, unlike individual tubes, plate-based PCR operates as a multi-well system where all reactions share a common physical structure. Under these conditions, small differences in plate behavior—such as how consistently it maintains contact, alignment, or structural stability—do not remain isolated. Instead, they can propagate across wells and appear as systematic variability that is difficult to interpret.
As a result, what seems to be a reagent issue or an instrument limitation may, in some cases, originate from factors related to the plate that are not immediately visible during routine setup. This is why some forms of variability are not random, but structured—repeating in ways that are noticeable in practice, yet difficult to explain. Recognizing this possibility is an important first step toward understanding why PCR results sometimes vary beyond what conventional assumptions would suggest.
Why PCR Variability Is Often Misdiagnosed
When PCR results vary, troubleshooting typically follows a familiar path, with reagents, pipetting, and instrument performance quickly becoming the usual suspects. Laboratories tend to begin by checking reagent integrity, reviewing pipetting consistency, and verifying thermal cycler conditions. This approach is logical, as these variables are directly involved in reaction setup and are known to influence outcomes. In many cases, these checks do resolve the issue. However, when they do not, the troubleshooting process often becomes repetitive rather than broader.
This is partly because the same logic that makes these variables easy to investigate also creates a blind spot. Factors that are considered “standard” or “fixed” are less likely to be questioned, even when they are part of the system influencing the result. PCR plates fall into this category. As consumables, they are generally assumed to be interchangeable as long as they meet basic specifications, and once a plate format has been selected, it is rarely revisited during troubleshooting unless there is an obvious compatibility issue.
In practice, this assumption works well when differences between plates are negligible. However, it becomes limiting when small variations in plate behavior begin to influence how reactions perform collectively. Because these effects are not tied to a single reaction but distributed across the plate, they are not easily recognized as a separate variable.
Another reason misdiagnosis occurs is that variability in plate-based PCR rarely presents as a clear failure. Reactions typically still amplify, but consistency is reduced. Replicates may diverge slightly, certain regions of the plate may behave differently, or data quality may shift gradually over time. These patterns are often familiar in routine work, but they are rarely treated as diagnostic signals. Instead, they are usually attributed to more familiar sources of variation, such as pipetting technique or minor reagent fluctuations—especially when there is no obvious failure to investigate.
As a result, troubleshooting efforts may continue to focus on variables that are easier to adjust or verify, while overlooking factors embedded in the physical setup of the experiment. Under these conditions, the underlying cause of variability can remain unresolved, even when standard optimization steps have been carefully applied.
What Makes Plate-Based PCR Inherently More Sensitive
A key difference between tube-based PCR and plate-based PCR lies in how reactions are physically organized. In tube-based setups, each reaction exists as an isolated unit, with its own physical boundary and contact conditions. Variations in one tube typically remain confined to that reaction, making inconsistencies easier to trace and correct. In contrast, PCR plates bring multiple reactions into a shared physical framework, where all wells are connected through the same structure and subjected to the same handling, sealing, and thermal cycling environment.
This shared structure introduces a different type of sensitivity. Instead of reacting independently, wells within a plate are influenced by factors that operate across the entire surface. For example, how evenly the plate sits on the thermal block, how consistently it maintains contact under the heated lid, or how uniformly pressure is distributed during sealing can all vary slightly across the plate. These differences are usually small, but because they affect multiple wells at once, they do not remain isolated. Instead, they can lead to coordinated changes in how groups of wells behave.
Another important aspect is scale. Plate-based workflows typically involve a larger number of reactions performed simultaneously, often under reduced reaction volumes. In such conditions, small physical differences become more consequential. Slight variations in contact with the heating block can influence local heat transfer, while minor differences in sealing contact can affect how well individual wells retain volume during cycling. These effects are often too subtle to prevent amplification, but they can shift reaction conditions enough to reduce consistency between wells.
Because of these characteristics, plate-based PCR is inherently more sensitive to system-level factors rather than isolated variables. In practice, this means that variability is more likely to emerge as patterns—such as consistent differences between regions of a plate—rather than as random outliers. The challenge is that these effects are not immediately visible during setup. They do not produce clear errors, but instead appear as small, repeatable differences that only become noticeable when results are compared across wells or across runs.
Common Experimental Patterns That Are Often Misinterpreted
In plate-based PCR workflows, variability rarely appears as a complete reaction failure. Instead, it more often presents as patterns—subtle inconsistencies that are noticeable in practice, but not immediately attributable to a single cause. These behaviors are familiar in routine laboratory work, yet they are frequently interpreted through the lens of more obvious variables such as reagents or pipetting technique, particularly when no clear alternative explanation is available.
One common observation is variability between replicate wells that are expected to behave identically. Even when reaction setup is carefully controlled, replicates may show small but noticeable differences in amplification behavior or signal intensity. In practical terms, a Ct standard deviation of around 0.2–0.3 is often considered acceptable in well-controlled qPCR runs, whereas values approaching or exceeding 0.5 may indicate underlying variability that warrants further investigation. Because these differences are often relatively small, they are frequently attributed to minor pipetting variation. However, when such variability appears consistently across certain groups of wells or repeats across plates, it is more likely to reflect differences in how physical conditions are maintained rather than simple dispensing error.
Another frequently encountered pattern is positional variation across the plate. Wells located near the edges or in specific regions may behave differently from those in the center, creating gradients or clusters of performance differences. In some cases, differences between edge and central wells can reach ΔCt values of approximately 0.3–1.0, depending on the system and conditions. While these effects are often associated with thermal cycler characteristics or environmental factors, consistent regional patterns are more likely to indicate that conditions such as contact, pressure, or sealing effectiveness are not uniform across the plate surface.
In qPCR workflows, these issues tend to become more visible in the data. Variability may appear as increased dispersion in Ct values, subtle shifts in amplification curves, or differences in signal consistency across wells prepared under the same conditions. While such variations are often attributed to reagent preparation or instrument sensitivity, their persistence under otherwise controlled conditions suggests that the source of variability may lie elsewhere in the system.
Because these patterns are neither random nor clearly linked to a single variable, they are often accepted as background variation rather than investigated further. In practice, many laboratories recognize these behaviors, but they are rarely treated as diagnostic signals—particularly when amplification still appears successful.
Why These Patterns Are Hard to Trace Back to Plate Design
One reason these patterns are difficult to attribute to PCR plate design is that plate-related effects rarely produce clear, isolated failures. Unlike reagent degradation or instrument malfunction—which can lead to obvious reaction loss and trigger immediate investigation—plate-related influences tend to modify how reactions perform rather than whether they work at all. Amplification still occurs, but consistency is affected in ways that are subtle, often remaining within an acceptable range and therefore easy to overlook.
Another challenge lies in how variability presents within a plate. Because plate-based PCR operates as a shared system, any structural or mechanical influence is distributed across multiple wells. This means that there is no single well that clearly “fails” in a way that points to the underlying cause. Instead, variability appears as spatially distributed differences—slight shifts across regions or gradual differences between replicates—which are inherently more difficult to diagnose than discrete errors and are often interpreted as normal experimental variation.
In addition, standard troubleshooting workflows are not designed to isolate plate-related variables. When issues arise, laboratories can readily test new reagents, recalibrate pipettes, or verify instrument settings—steps that provide immediate feedback. In contrast, evaluating the impact of plate design is less straightforward. Differences between plates are often subtle, and unless side-by-side comparisons are performed under controlled conditions, their effects can easily be masked by more familiar sources of variation. In practice, this means that plate-related issues are often not tested directly, but instead inferred only after other possibilities have been exhausted.
There is also a tendency to interpret variability through variables that are easier to conceptualize. Reagents, pipetting, and thermal cycling are directly linked to reaction chemistry, making them intuitive points of focus during troubleshooting. Plate design, on the other hand, influences the physical environment in which reactions occur—through contact, alignment, and structural behavior—which is less visible during setup and therefore less likely to be questioned. As a result, its contribution to variability is often considered only when more obvious explanations fail to account for the observed patterns.
Taken together, these factors create a situation in which plate-related variability can persist across experiments without being clearly identified. The absence of obvious failure, combined with the distributed and subtle nature of the effects, makes it possible for variability to be repeatedly observed, but not fully explained—something many laboratories recognize, even if the underlying cause is not immediately clear.
The Hidden Role of PCR Plate Design
Variability in PCR plate workflows cannot always be fully explained by reagents, pipetting, or instrument settings, it becomes necessary to examine how the physical environment of the reaction is structured. In plate-based PCR, this environment is defined by the plate itself. Rather than acting as a passive container, the PCR plate provides the mechanical and structural framework within which all reactions take place simultaneously. In practice, differences in plate design can influence how uniformly conditions are maintained across wells, which is why selecting appropriate PCR plates becomes an important consideration in achieving consistent results.
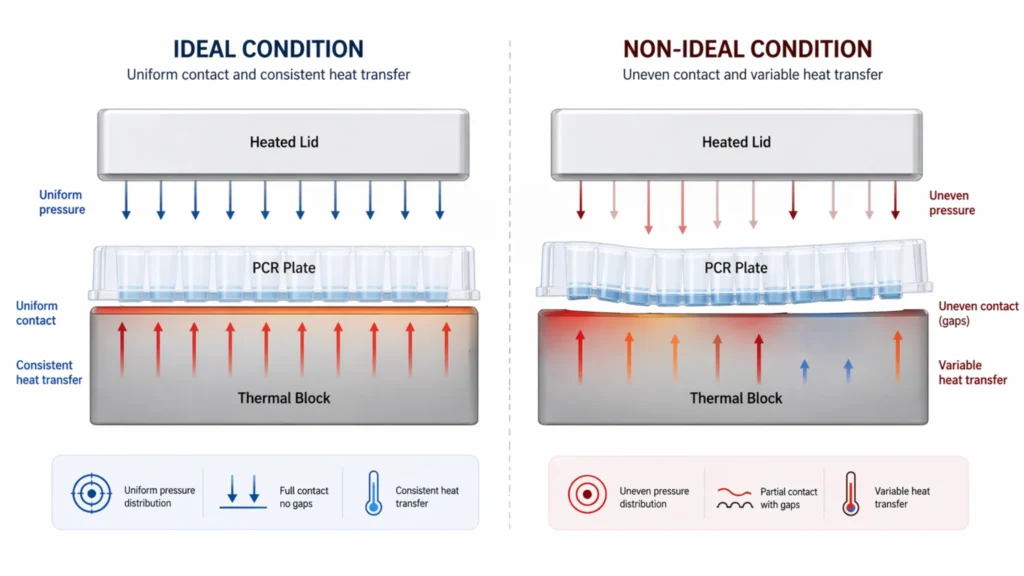
One of the key roles of plate design is to maintain consistent structural support across all wells. In practice, even small differences in rigidity or flatness can influence how evenly the plate sits on the thermal block and how consistently it maintains contact under the heated lid. If certain areas of the plate have slightly less support or contact, those wells may experience subtle differences in heat transfer efficiency or lid pressure. Over repeated thermal cycles, even minor differences in thermal contact resistance can lead to measurable divergence in reaction behavior between wells. These differences are usually too small to disrupt amplification entirely, but they can reduce consistency across the plate.
Plate design also contributes to how uniformly conditions are maintained during routine handling and processing. Plates are typically moved, sealed, and sometimes centrifuged as a single unit, which means that any structural flexibility is expressed across multiple wells at once. For example, slight bending during handling or uneven pressure during sealing can alter how tightly certain regions are sealed or supported. During thermal cycling, this can translate into differences in evaporation control or local reaction conditions, particularly in regions where sealing contact is less consistent.
Another important aspect is uniformity at scale. In a multi-well format, consistency is not determined by whether individual wells function, but by how similar all wells are under the same conditions. Plate design influences this by determining how evenly physical forces—such as contact with the heating block, lid pressure, and sealing contact—are distributed across the entire surface. When this distribution is uneven, small physical differences can accumulate and become visible as structured variability, such as reproducible differences between regions of the plate.
For this reason, the impact of PCR plate design is often indirect. It does not introduce a single, easily identifiable variable, but instead modifies how existing variables—such as heat transfer, sealing performance, and mechanical stability—are experienced across the system. In plate-based PCR, consistency is not only a chemical outcome, but also a physical one. This is why plate-related effects are often subtle yet persistent, and why they tend to appear as patterns rather than as isolated inconsistencies.
How Plate Design Interacts with Other Variables
In plate-based PCR workflows, variability is rarely the result of a single factor acting in isolation. Instead, it often emerges from how multiple variables interact within the same physical system. Plate design plays a central role in this interaction, not because it dominates the reaction, but because it determines how consistently other variables—such as sealing, heating, and handling—are experienced across the plate.
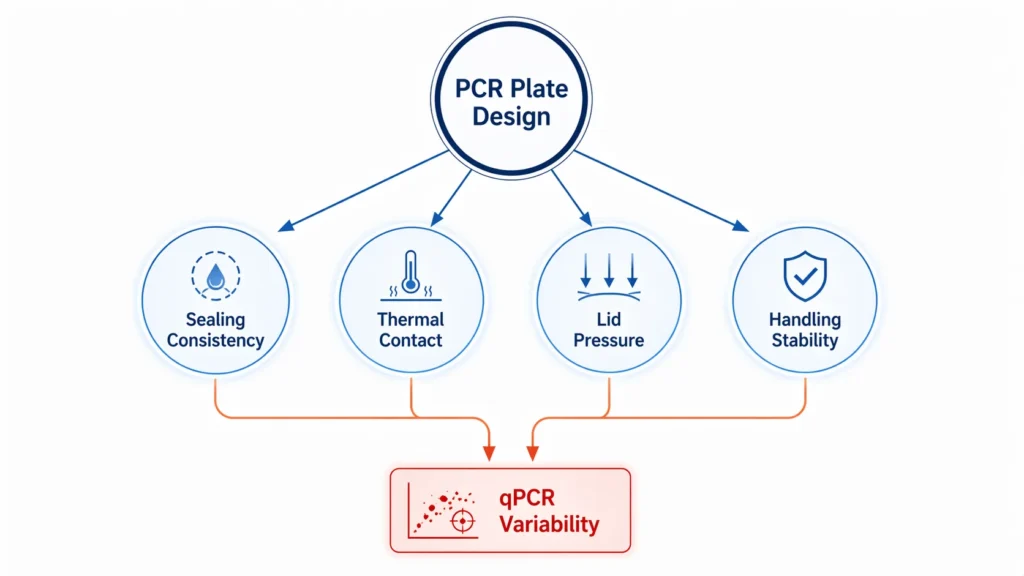
One of the most common interactions occurs between the plate and the sealing process. While sealing is intended to create a uniform barrier across all wells, its effectiveness depends on how evenly the plate surface supports contact. In practice, both the sealing material and the underlying plate structure contribute to overall performance, making sealing consistency an important factor to consider in routine workflows. For example, if certain areas of the plate sit slightly higher or lower due to minor differences in flatness or rigidity, the sealing film may not contact all wells with the same pressure. In practice, this can lead to tighter sealing in some regions and weaker sealing in others, resulting in uneven evaporation control during thermal cycling.
A similar interaction can be observed between the plate and the thermal cycler. Thermal cyclers are designed to provide consistent heating and lid pressure, but this consistency assumes that the plate maintains uniform contact with both the heating block and the lid. If the plate does not sit evenly, or if certain areas flex slightly under lid pressure, heat transfer and pressure distribution can vary across wells. As a result, differences in amplification behavior may appear across regions of the plate, even when the instrument itself is functioning within specifications.
Handling introduces another layer of interaction. During routine workflows, plates are moved, sealed, and sometimes centrifuged as a single unit. If the plate has limited structural stability, even small deformations during these steps can alter alignment or contact conditions before cycling begins. These changes are typically not visible during setup, but they can influence how consistently wells behave during the reaction, particularly when combined with sealing and thermal cycling effects.
These interactions share a common characteristic: they do not create entirely new sources of variability, but instead amplify small physical differences that already exist within the system. Each component—sealing, thermal cycling, handling—may function within acceptable limits on its own. However, when combined with variations in plate behavior, their effects can diverge across the plate in a coordinated way. This is why variability can still emerge in a coordinated manner, even when all individual steps appear to be performed correctly.
Understanding PCR variability in this way shifts the focus from isolating single causes to recognizing patterns of interaction. Rather than asking which variable is responsible, it becomes more useful to consider how variables reinforce or amplify each other within the shared structure of the plate. This perspective helps explain why certain issues persist despite standard optimization and why adjusting a single parameter does not always lead to consistent improvement.
A Practical Way to Re-Evaluate PCR Variability
When variability persists despite standard optimization, it can be useful to step back and reconsider how the system is being evaluated. In many cases, troubleshooting focuses on variables that are easiest to isolate and adjust, such as reagents, pipetting technique, or instrument settings. While this approach is effective for addressing obvious issues, it may not fully capture factors that operate at the level of the overall system—particularly when variability remains after multiple rounds of adjustment.
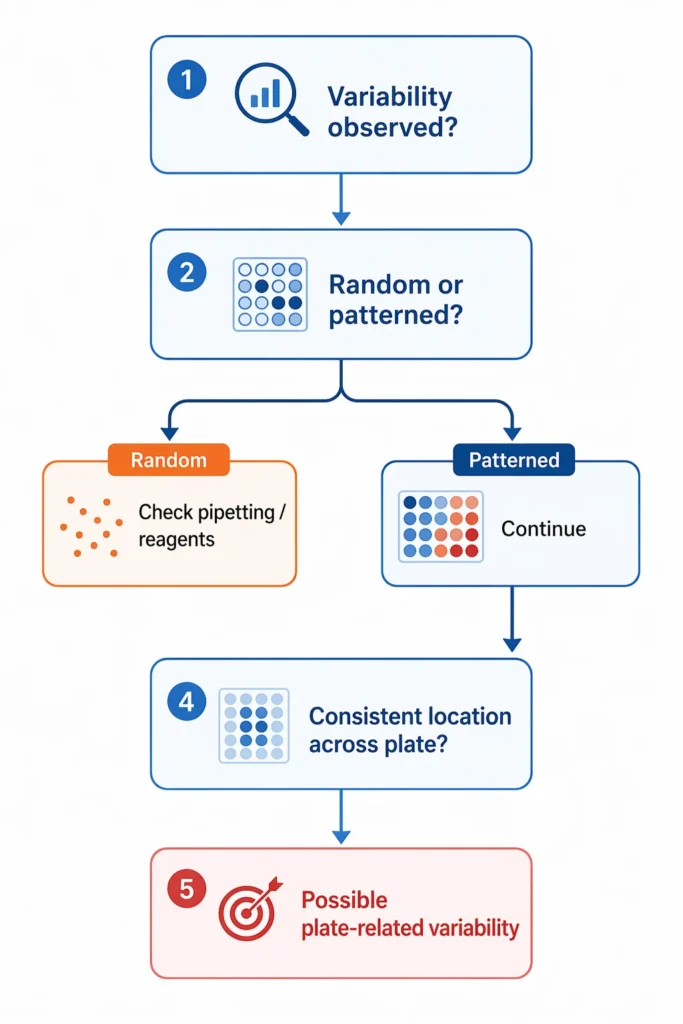
A more effective approach is to examine variability in terms of patterns rather than isolated events. For example, differences that consistently appear in specific regions of a plate, or variability that repeats across runs under otherwise stable conditions, often indicate that the source of the issue is not random. In practice, these patterns can serve as useful diagnostic signals, suggesting that physical conditions across the plate may not be as uniform as assumed.
In this context, it becomes relevant to look more closely at how the workflow is physically implemented. This may involve observing whether the plate sits evenly in the thermal cycler, whether sealing is applied consistently across the entire surface, or whether certain regions of the plate are more sensitive during handling. These aspects are often treated as fixed once a workflow is established, but revisiting them can sometimes reveal sources of variability that are not apparent from protocol alone.
Another useful perspective is to evaluate consistency at the system level rather than at the level of individual components. A workflow in which each step appears to function correctly in isolation may still produce variability if interactions between components are not fully aligned. In such cases, simple comparisons—such as testing different plate batches or suppliers or observing how variability changes under slightly different handling conditions—can provide more insight than repeatedly adjusting reagents or cycling parameters.
Ultimately, re-evaluating PCR variability involves expanding the scope of troubleshooting. Instead of focusing only on variables that directly affect reaction chemistry, it can be helpful to also consider how the physical environment of the reaction influences consistency across the system. This broader perspective does not replace standard optimization, but it can complement it by identifying sources of variability that are otherwise easy to overlook.
Practical Signals to Identify Plate-Related Variability
While plate-related variability is not always easy to isolate, certain recurring patterns can provide useful clues when interpreting inconsistent PCR results.
A key signal is spatial consistency. If variability repeatedly appears in the same regions of a plate—such as edge wells behaving differently from central wells, or specific rows showing higher dispersion across runs—it is unlikely to be caused by random pipetting error. Instead, such patterns more strongly suggest that physical conditions, such as contact with the thermal block, lid pressure distribution, or sealing uniformity, are not evenly maintained across the plate.
Another important observation is persistence despite optimization. When reagent conditions, pipetting technique, and instrument settings have been carefully adjusted, but the overall pattern of variability remains similar, the source of the issue is unlikely to lie in these variables alone. In practice, this is often the point at which further optimization yields diminishing returns, and system-level factors—particularly those affecting multiple wells simultaneously—become more relevant.
Comparative testing can also provide insight. Running the same protocol using plates from a different batch or supplier, or observing how variability changes under slightly modified handling or sealing conditions, can help reveal whether plate behavior is influencing results. If the pattern shifts with the plate rather than with the protocol, this is a strong indication that plate-related factors are involved.
Taken together, these observations do not provide a definitive diagnosis, but they help narrow the scope of troubleshooting. Rather than treating variability as random or unavoidable, they allow it to be interpreted as a structured signal—one that may reflect how consistently physical conditions are maintained across the plate. In practice, recognizing these signals can help avoid repeated adjustments to the wrong variables, and instead guide attention toward factors that influence the system as a whole.
Conclusion
Variability in PCR and qPCR workflows is often approached as a problem of reagents, pipetting, or instrument performance. While these factors are critical, they do not always provide a complete explanation for the patterns observed in plate-based experiments. As discussed throughout this article, some forms of variability originate not from a single source, but from how reactions are physically organized and how different elements of the workflow interact within that structure.
PCR plates, as part of the physical system, influence how consistently conditions are maintained across wells. Their role is not always obvious because their effects are typically indirect—modifying how other variables are experienced rather than introducing new ones in isolation. In practice, this means that variability in plate-based PCR is often structured rather than random, even when it is initially interpreted as routine fluctuation.
Recognizing the potential contribution of plate design does not replace conventional optimization strategies, but it expands the way variability is understood and addressed. Considering both the chemical and physical aspects of the workflow allows for a more complete interpretation of experimental results, particularly when inconsistencies persist without a clear cause.
For laboratories working with plate-based PCR systems, revisiting how plate behavior aligns with handling methods, sealing approaches, and instrument conditions can provide valuable insight. In some cases, comparing different plate types or suppliers, or observing how variability changes under slightly modified conditions, can reveal patterns that are otherwise difficult to explain. This broader perspective helps move troubleshooting beyond repeated adjustment and toward a more systematic understanding of reproducibility.
FAQ: Understanding PCR Plate-Related Variability
What level of Ct variation is considered acceptable in qPCR?
In well-controlled qPCR experiments, a Ct standard deviation of approximately 0.2–0.3 is generally considered acceptable. Values approaching or exceeding 0.5 may indicate increased variability that warrants further investigation, particularly if consistent patterns are observed.
How can I tell if variability is caused by the PCR plate rather than pipetting?
Variability that appears randomly across wells is more often associated with pipetting or reagent factors. In contrast, variability that shows consistent spatial patterns—such as differences between edge and central wells, or repeated variation in specific regions across runs—is more likely to be linked to plate-related physical conditions.
Can changing PCR plates improve reproducibility?
In some cases, yes. If variability persists despite optimization of reagents and instrument settings, comparing results using plates from different batches or suppliers can help determine whether plate-related factors are contributing to inconsistency.
Why does variability often appear at the edges of a PCR plate?
Edge wells may be more sensitive to differences in thermal contact, lid pressure, or sealing consistency. These factors can vary slightly across the plate surface, leading to more noticeable variability in peripheral regions.
Is plate-related variability more important in qPCR than in standard PCR?
Yes. Because qPCR measures amplification in real time, it is more sensitive to small differences in reaction conditions. Variability that may not be obvious in endpoint PCR can become more apparent in qPCR data.
If you are working with PCR plates in routine laboratory workflows, evaluating how sealing consistency, plate compatibility, and application methods align with your experimental setup is often more informative than relying on specifications alone.
Reviewing commonly used PCR plate and sealing configurations can provide a practical point of reference when comparing different approaches or validating potential sources of variability in both standard PCR and qPCR workflows.
📩 For pricing information, sample requests, or configuration consultation, please feel free to contact us.


