Plate sealing is often treated as a simple closing step in PCR workflows, mainly intended to prevent evaporation during thermal cycling. In practice, however, the quality and consistency of sealing can directly influence how stable and reproducible PCR and qPCR results are, especially when working with multi-well plates. For a broader overview of how plate formats and consumables affect PCR performance, see our guide on PCR plates and related workflows.
Because sealing is usually performed after reaction setup and before cycling begins, its impact is not always immediately visible. Variations in sealing pressure, film fit, or plate compatibility can introduce subtle differences across wells, which may later appear as inconsistent amplification, increased variability, or edge effects that are difficult to explain through reagents or instrument settings alone.
For this reason, plate sealing should be considered not just as a procedural step, but as part of the overall system that determines reaction consistency in plate-based PCR workflows.
Why sealing becomes a critical variable in plate-based PCR workflows
In plate-based PCR workflows, sealing does more than simply close the reaction environment. Unlike individual tubes, where each reaction is physically separated, PCR plates introduce an additional layer of complexity: multiple reactions are performed simultaneously within a shared structure, often under low-volume conditions. This makes the system inherently more sensitive to small inconsistencies.
One of the key differences is that sealing is applied across the entire plate rather than on a per-reaction basis. In practice, this means that any variation in pressure, alignment, or film contact is not isolated to a single reaction, but distributed unevenly across multiple wells. As a result, what appears to be a minor imperfection at the sealing stage can translate into measurable variability between wells during thermal cycling.
This effect becomes more pronounced in routine laboratory work where sealing is performed manually or under time constraints. Small differences in how the film is applied — for example, uneven pressure at the edges or slight misalignment — can lead to localized differences in sealing integrity. These differences are rarely obvious during setup, but they tend to emerge later as inconsistent amplification patterns, particularly when comparing edge wells to central wells. In such cases, using a well-designed PCR sealing film that supports consistent contact across the plate can help reduce variability introduced during this step.
For this reason, sealing should be viewed as part of the overall system that governs reaction uniformity in PCR plates. Its role is not only to prevent evaporation, but to maintain consistent conditions across all wells, which is essential for reliable and comparable results in multi-well experiments.
Evaporation control starts with sealing quality
In PCR plate workflows, evaporation is often discussed as a thermal cycling issue, but in practice it is closely linked to how well the plate is sealed before cycling even begins. When sealing is not uniform across the plate, differences in vapor retention can develop across wells, leading to localized volume loss during thermal cycling.
Even small changes in reaction volume can alter the effective concentration of reagents within a well. Similar effects have also been discussed in relation to PCR tube performance during thermal cycling, where reaction stability depends on consistent volume and sealing conditions. In PCR, and especially in low-volume reactions, this can influence amplification efficiency without necessarily causing complete reaction failure. Instead, the impact is more often reflected in reduced consistency across wells rather than in obvious reaction loss.
In routine laboratory work, these effects are frequently associated with positional differences across the plate. While such patterns are often attributed to temperature gradients within the thermal cycler, uneven sealing can further amplify these effects by allowing greater evaporation in certain regions. As a result, what appears to be an instrument-related issue may, in fact, originate from inconsistencies introduced during the sealing step.
From a practical standpoint, evaporation-related variability is not always immediately visible after cycling. It is more often identified through overall data consistency — for example, when expected uniform reactions show unexplained dispersion. Recognizing this connection is important, because it shifts troubleshooting from focusing solely on instruments or reagents toward evaluating sealing quality as part of the workflow.
Uneven sealing can create well-to-well variability across the plate
In plate-based PCR, the goal is not only to maintain each individual reaction, but also to ensure that all wells experience as similar conditions as possible. While evaporation affects overall reaction stability, uneven sealing introduces a different type of issue — variability between wells within the same plate.
Unlike individual tubes, where sealing is applied independently, PCR plates rely on a single sealing layer across all wells. In practice, this means that sealing quality is rarely perfectly uniform. Minor differences in pressure distribution, film contact, or plate flatness can result in certain regions of the plate being sealed more tightly than others. These differences are typically too subtle to detect during setup, but they can influence how each well behaves during thermal cycling.
The impact of this variability is often observed as inconsistent performance between replicates that are expected to behave identically. For example, wells located in the same region may show similar deviations, while others remain stable, creating patterns that are spatial rather than random. This type of inconsistency is particularly difficult to troubleshoot, because it does not necessarily correlate with reagent preparation or pipetting accuracy.
In qPCR applications, where data interpretation depends on small differences in signal, well-to-well variability introduced by uneven sealing can significantly affect result reliability. Even when amplification appears successful across all wells, increased dispersion between replicates or subtle shifts in signal curves may indicate that reaction conditions were not fully uniform across the plate.
For this reason, uneven sealing should be considered as a potential source of intra-plate variability, especially when experimental results show positional patterns that cannot be explained by other factors. Addressing this issue requires not only careful sealing technique, but also an understanding that consistency across the entire plate is as important as the performance of any single well.
How sealing affects contamination risk during and after setup
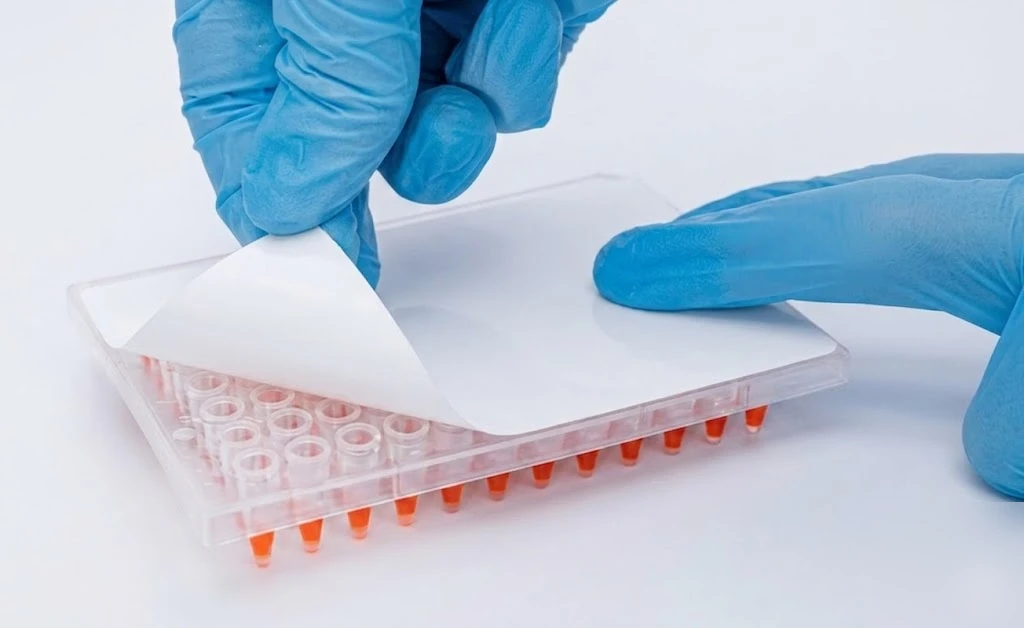
In plate-based PCR workflows, sealing does not only serve to stabilize reaction conditions, but also acts as a physical barrier that separates reactions after setup is completed. Once samples and reagents have been dispensed across the plate, the integrity of this barrier becomes critical, particularly in workflows involving multiple samples, replicates, or high-throughput processing.
When sealing is incomplete or uneven, the separation between wells is no longer fully reliable. During handling, transport, or even minor disturbances after setup, small amounts of liquid or aerosol can move between wells, especially if the sealing surface is not uniformly secured. Unlike in individual tubes, where contamination is typically confined to a single reaction, plate-based formats increase the risk that such events affect multiple wells simultaneously.
This risk is further influenced by how the plate is handled after sealing. In routine laboratory work, plates may be briefly repositioned, transferred between workstations, or subjected to vibration before thermal cycling begins. If the seal is not sufficiently robust, these steps can introduce cross-well contamination that is difficult to detect at the time but may later appear as unexpected amplification signals or inconsistent negative controls.
In addition, workflows that involve post-sealing manipulation — such as piercing, partial resealing, or extended handling — place additional demands on sealing integrity. Under these conditions, even small weaknesses in the initial seal can become points where contamination is introduced or propagated.
From a practical perspective, contamination related to sealing is often underestimated because it does not always produce obvious failures. Instead, it may appear as low-level background signals, sporadic positive results in control wells, or inconsistencies that are difficult to attribute to a single source. Recognizing sealing as part of contamination control helps shift attention toward handling steps that are otherwise considered routine, but can have significant impact on result reliability.
Why sealing matters even more in qPCR applications
In standard PCR workflows, small variations introduced during setup may not always lead to immediately visible differences in outcome. As long as amplification occurs and the final product is detectable, minor inconsistencies can remain unnoticed. However, qPCR operates under a different level of sensitivity, where even small changes in reaction conditions can influence how results are interpreted.
Because qPCR relies on real-time fluorescence measurement, it is inherently more sensitive to differences in reaction volume, concentration, and overall reaction environment. When sealing is uneven, the resulting differences between wells — whether caused by slight evaporation, pressure variation, or inconsistent film contact — can affect signal consistency across the plate.
These effects are often subtle rather than catastrophic. Reactions may still amplify successfully, but the overall consistency of the data can be reduced, making it more difficult to interpret results with confidence, particularly when comparing replicates or multiple samples.
One of the challenges in qPCR troubleshooting is that this type of variability is not always immediately attributed to sealing. It is often first associated with reagent preparation, pipetting accuracy, or instrument performance. However, when such factors have been controlled and variability persists in a positional or pattern-based manner, sealing becomes an important variable to consider.
For this reason, the role of sealing in qPCR workflows extends beyond simply preventing evaporation. It becomes part of the broader system that supports consistent and interpretable data, especially in experiments that rely on precise comparison across wells.
Common signs that sealing may be affecting your results
In routine PCR and qPCR work, issues related to sealing rarely present as complete reaction failure. Instead, they tend to appear as subtle patterns in the data, which are often attributed to other factors unless specifically examined.
One of the most common indicators is positional variability across the plate. When wells located in similar regions — such as edges or corners — consistently behave differently from central wells, it may suggest that sealing is not uniform across the plate. This type of pattern is particularly important because it reflects spatial differences rather than random variation.
Another sign is increased dispersion between replicate wells that are otherwise prepared under identical conditions. When pipetting accuracy and reagent preparation have been controlled, but replicates still show unexpected variability, uneven sealing should be considered as a possible contributing factor.
In qPCR workflows, these patterns often become more apparent in the data. Variability may appear as shifts in Ct values, changes in amplification curve profiles, or inconsistent signal intensity between wells that are expected to behave similarly.
Changes in reaction volume after thermal cycling can also provide indirect evidence. Although not always easy to observe, slight differences in liquid levels between wells may indicate that evaporation has occurred unevenly, often as a result of inconsistent sealing. In practice, this is more likely to be noticed when comparing wells side by side, rather than evaluating individual reactions in isolation.
Unexpected signals in negative controls or low-level background amplification may also be associated with sealing-related issues, particularly when they occur sporadically or in specific regions of the plate. While contamination is often the first explanation, it is important to consider whether sealing integrity during or after setup may have contributed to cross-well interference.
Taken together, these signs are rarely definitive on their own. However, when multiple patterns — such as positional effects, replicate variability, and unexplained signal behavior — appear simultaneously, they provide a strong indication that sealing should be evaluated as part of the overall workflow.
What to consider when choosing a sealing solution for PCR plates
Selecting a sealing solution for PCR plates is not simply a matter of choosing a material, but of ensuring that the sealing approach is compatible with the specific workflow and performance requirements of the experiment. In practice, sealing performance depends on how consistently the seal can be applied across the entire plate, rather than on any single property of the sealing film or mat.
One of the primary considerations is the compatibility between the sealing material and the plate itself. Differences in plate rigidity, well geometry, or surface finish can influence how evenly a sealing film adheres. A combination that performs well in one setup may not produce the same level of consistency in another, particularly when different plate formats or manufacturers are involved.
Consistency of application is equally important. In many laboratories, sealing is performed manually, which introduces variability in how pressure is distributed across the plate surface. Even with the same sealing material, differences in application technique can lead to uneven contact between the film and individual wells. For this reason, the choice of sealing solution should take into account not only material properties, but also how it will be applied in routine use.
For workflows involving qPCR or other sensitive applications, optical compatibility becomes an additional factor. Sealing materials that interfere with fluorescence detection, or that do not maintain consistent optical clarity across the plate, can introduce variability in signal measurement. In such cases, the sealing approach must support both physical isolation and reliable signal acquisition.
It is also important to consider how the plate will be handled after sealing. Workflows that involve transport, extended handling, or additional processing steps place greater demands on sealing stability. Under these conditions, a sealing solution that maintains integrity under routine movement and manipulation is more likely to preserve consistent reaction conditions across all wells.
Ultimately, the effectiveness of a sealing solution is best evaluated in terms of how well it supports uniform conditions across the entire plate throughout the workflow. Rather than focusing solely on individual features, it is more useful to consider whether the sealing approach can reliably minimize variability under the specific conditions of use.
A practical checklist for more reliable plate sealing
To maintain consistent conditions across PCR plates, sealing should be approached as a controlled step rather than a routine action. In practice, a few key considerations can significantly reduce variability and improve overall result reliability.
- Confirm compatibility between plate and sealing material
Ensure that the sealing solution matches the plate format and surface characteristics, as mismatches can lead to uneven adhesion. - Apply sealing evenly across the entire plate surface
Consistent pressure distribution is critical. Uneven application can result in localized differences in sealing integrity that affect individual wells. - Pay particular attention to peripheral wells in low-volume reactions
Edge regions are more susceptible to variability, and sealing inconsistencies in these areas are more likely to influence results. - Consider optical requirements for qPCR workflows
Sealing materials should support reliable fluorescence detection without introducing signal distortion or variability. - Evaluate sealing performance based on data patterns, not appearance alone
A film that appears well-attached may still produce variability if sealing is not uniform across all wells. - Review handling steps after sealing
Movement, repositioning, or additional processing can compromise sealing integrity if not properly managed.
Taken together, these practices help ensure that sealing supports consistent reaction conditions across all wells, reducing the likelihood that variability originates from factors outside the intended experimental design. For laboratories standardizing their workflows, selecting compatible PCR plates and sealing solutions can further improve consistency across experiments.
Reliable PCR results depend on more than the thermal cycler program
In plate-based PCR and qPCR workflows, result consistency is often attributed to factors such as reagent quality or instrument performance. While these are undoubtedly important, the role of sealing is frequently underestimated. As a step that directly influences reaction stability, uniformity, and isolation across wells, sealing forms part of the foundation on which reliable results are built.
When sealing is considered as an integral part of the workflow — rather than a routine final step — many sources of unexplained variability become easier to identify and control. For laboratories evaluating different plate and sealing combinations, variability that appears to originate from reagents or instruments can often be reduced by improving sealing consistency within the workflow.
Frequently Asked Questions About PCR Plate Sealing
Does plate sealing affect PCR results even if amplification appears normal?
Yes. Sealing-related issues do not always cause complete reaction failure. In many cases, amplification still occurs, but variability between wells increases. This can lead to less consistent results, particularly in qPCR where small differences in Ct values are significant.
Why do edge wells behave differently in PCR plates?
Edge wells are more sensitive to environmental variation, including temperature gradients and evaporation. If sealing is not uniform across the plate, these effects can be amplified at the periphery, leading to differences compared to central wells.
Can uneven sealing be mistaken for pipetting or reagent problems?
Yes, this is a common situation. Variability caused by uneven sealing is often first attributed to pipetting accuracy or reagent preparation. However, when inconsistencies show positional patterns across the plate, sealing should be considered as a potential contributing factor.
How can you tell if sealing is causing variability in qPCR data?
Typical indicators include increased dispersion between replicates, consistent differences between specific regions of the plate, or unexpected variation in Ct values despite controlled experimental conditions. These patterns often suggest that reaction conditions were not uniform across all wells.
If you are working with PCR plates in routine laboratory workflows, focusing on how sealing consistency, plate compatibility, and application methods align with your experimental setup is often more practical than relying on specifications alone. For laboratories evaluating suitable configurations, our PCR plate and sealing solutions provide a useful reference for standard PCR and qPCR applications.
📩 For pricing information, sample requests, or configuration consultation, please feel free to contact us.


